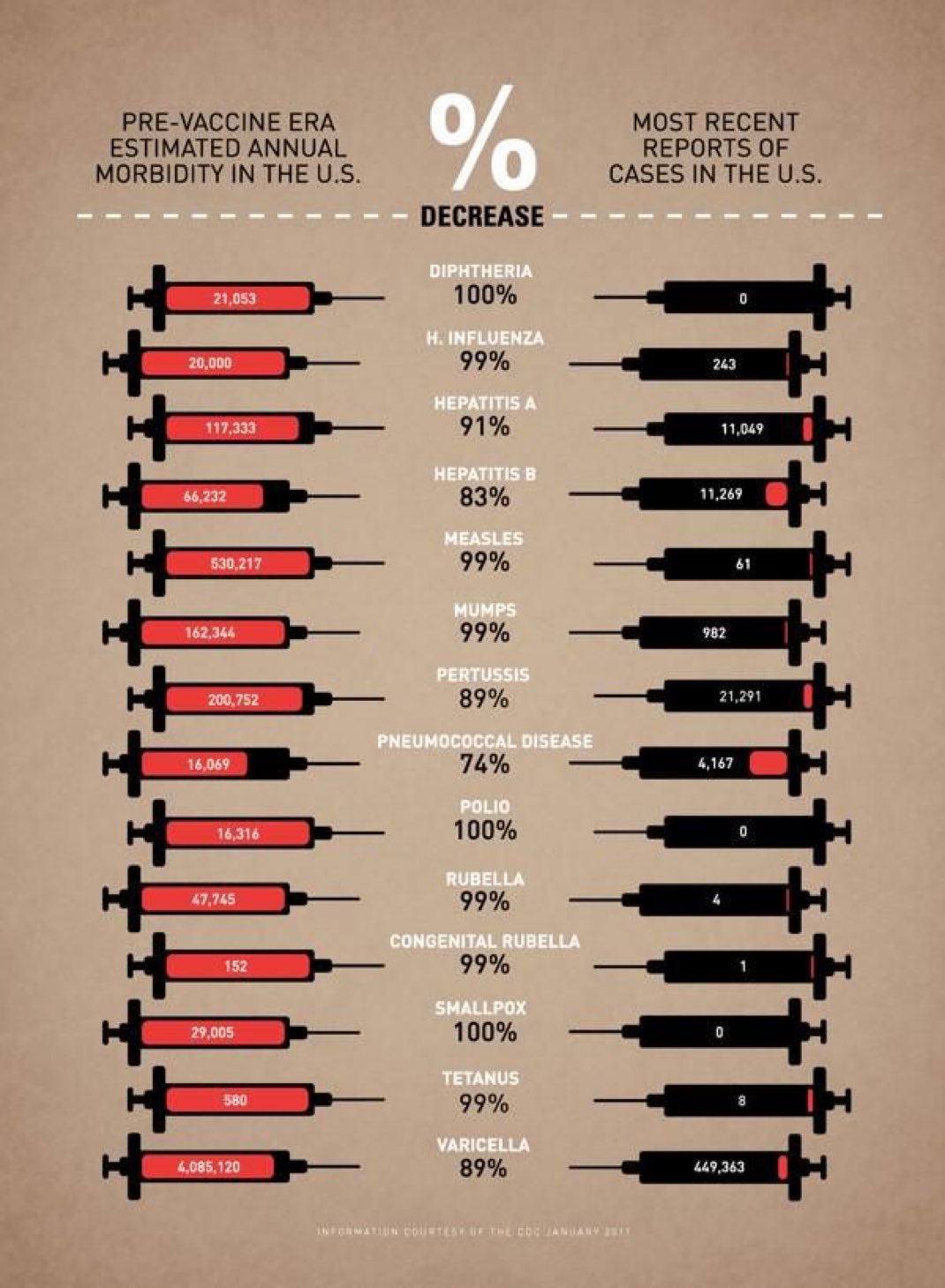
Malaria is caused by Plasmodium parasites. The parasites are spread to people through the bites of infected female Anopheles mosquitoes, called “malaria vectors.” There are 5 parasite species that cause malaria in humans, and 2 of these species – P. falciparum and P. vivax – pose the greatest threat.
In 2018, P. falciparum accounted for 99.7% of estimated malaria cases in the WHO African Region 50% of cases in the WHO South-East Asia Region, 71% of cases in the Eastern Mediterranean and 65% in the Western Pacific.
P. vivax is the predominant parasite in the WHO Region of the Americas, representing 75% of malaria cases.
Symptoms
Malaria is an acute febrile illness. In a non-immune individual, symptoms usually appear 10–15 days after the infective mosquito bite. The first symptoms – fever, headache, and chills – may be mild and difficult to recognize as malaria. If not treated within 24 hours, P. falciparum malaria can progress to severe illness, often leading to death.
Children with severe malaria frequently develop one or more of the following symptoms: severe anaemia, respiratory distress in relation to metabolic acidosis, or cerebral malaria. In adults, multi-organ failure is also frequent. In malaria endemic areas, people may develop partial immunity, allowing asymptomatic infections to occur.
Who is at risk?
In 2019, nearly half of the world’s population was at risk of malaria. Most malaria cases and deaths occur in sub-Saharan Africa. However, the WHO regions of South-East Asia, Eastern Mediterranean, Western Pacific, and the Americas are also at risk.
Some population groups are at considerably higher risk of contracting malaria, and developing severe disease, than others. These include infants, children under 5 years of age, pregnant women and patients with HIV/AIDS, as well as non-immune migrants, mobile populations and travellers. National malaria control programmes need to take special measures to protect these population groups from malaria infection, taking into consideration their specific circumstances.
Disease burden
According to the latest World malaria report, released on 30 November 2020, there were 229 million cases of malaria in 2019 compared to 228 million cases in 2018. The estimated number of malaria deaths stood at 409 000 in 2019, compared with 411 000 deaths in 2018.
The WHO African Region continues to carry a disproportionately high share of the global malaria burden. In 2019, the region was home to 94% of all malaria cases and deaths.
In 2019, 6 countries accounted for approximately half of all malaria deaths worldwide: Nigeria (23%), the Democratic Republic of the Congo (11%), United Republic of Tanzania (5%), Burkina Faso (4%), Mozambique (4%) and Niger (4% each).
Children under 5 years of age are the most vulnerable group affected by malaria; in 2019 they accounted for 67% (274 000) of all malaria deaths worldwide.
Transmission
In most cases, malaria is transmitted through the bites of female Anopheles mosquitoes. There are more than 400 different species of Anopheles mosquito; around 30 are malaria vectors of major importance. All of the important vector species bite between dusk and dawn. The intensity of transmission depends on factors related to the parasite, the vector, the human host, and the environment.
Anopheles mosquitoes lay their eggs in water, which hatch into larvae, eventually emerging as adult mosquitoes. The female mosquitoes seek a blood meal to nurture their eggs. Each species of Anopheles mosquito has its own preferred aquatic habitat; for example, some prefer small, shallow collections of fresh water, such as puddles and hoof prints, which are abundant during the rainy season in tropical countries.
Transmission is more intense in places where the mosquito lifespan is longer (so that the parasite has time to complete its development inside the mosquito) and where it prefers to bite humans rather than other animals. The long lifespan and strong human-biting habit of the African vector species is the main reason why approximately 90% of the world’s malaria cases are in Africa.
Transmission also depends on climatic conditions that may affect the number and survival of mosquitoes, such as rainfall patterns, temperature and humidity. In many places, transmission is seasonal, with the peak during and just after the rainy season. Malaria epidemics can occur when climate and other conditions suddenly favour transmission in areas where people have little or no immunity to malaria. They can also occur when people with low immunity move into areas with intense malaria transmission, for instance to find work, or as refugees.
Human immunity is another important factor, especially among adults in areas of moderate or intense transmission conditions. Partial immunity is developed over years of exposure, and while it never provides complete protection, it does reduce the risk that malaria infection will cause severe disease. For this reason, most malaria deaths in Africa occur in young children, whereas in areas with less transmission and low immunity, all age groups are at risk.
Prevention
Vector control is the main way to prevent and reduce malaria transmission. If coverage of vector control interventions within a specific area is high enough, then a measure of protection will be conferred across the community.
WHO recommends protection for all people at risk of malaria with effective malaria vector control. Two forms of vector control – insecticide-treated mosquito nets and indoor residual spraying – are effective in a wide range of circumstances.
Insecticide-treated mosquito nets
Sleeping under an insecticide-treated net (ITN) can reduce contact between mosquitoes and humans by providing both a physical barrier and an insecticidal effect. Population-wide protection can result from the killing of mosquitoes on a large scale where there is high access and usage of such nets within a community.
In 2019, an estimated 46% of all people at risk of malaria in Africa were protected by an insecticide-treated net, compared to 2% in 2000. However, ITN coverage has been at a standstill since 2016.Indoor spraying with residual insecticides
Indoor residual spraying (IRS) with insecticides is another powerful way to rapidly reduce malaria transmission. It involves spraying the inside of housing structures with an insecticide, typically once or twice per year. To confer significant community protection, IRS should be implemented at a high level of coverage.
Globally, IRS protection declined from a peak of 5% in 2010 to 2% in 2019, with decreases seen across all WHO regions, apart from the WHO Eastern Mediterranean Region. The declines in IRS coverage are occurring as countries switch from pyrethroid insecticides to more expensive alternatives to mitigate mosquito resistance to pyrethroids.
Antimalarial drugs
Antimalarial medicines can also be used to prevent malaria. For travellers, malaria can be prevented through chemoprophylaxis, which suppresses the blood stage of malaria infections, thereby preventing malaria disease. For pregnant women living in moderate-to-high transmission areas, WHO recommends at least 3 doses of intermittent preventive treatment with sulfadoxine-pyrimethamine at each scheduled antenatal visit after the first trimester. Similarly, for infants living in high-transmission areas of Africa, 3 doses of intermittent preventive treatment with sulfadoxine-pyrimethamine are recommended, delivered alongside routine vaccinations.
Since 2012, WHO has recommended seasonal malaria chemoprevention as an additional malaria prevention strategy for areas of the Sahel sub-region of Africa. The strategy involves the administration of monthly courses of amodiaquine plus sulfadoxine-pyrimethamine to all children under 5 years of age during the high transmission season.
Insecticide resistance
Since 2000, progress in malaria control has resulted primarily from expanded access to vector control interventions, particularly in sub-Saharan Africa. However, these gains are threatened by emerging resistance to insecticides among Anopheles mosquitoes. According to the latest World malaria report, 73 countries reported mosquito resistance to at least 1 of the 4 commonly-used insecticide classes in the period 2010-2019. In 28 countries, mosquito resistance was reported to all of the main insecticide classes.
Despite the emergence and spread of mosquito resistance to pyrethroids, insecticide-treated nets continue to provide a substantial level of protection in most settings. This was evidenced in a large 5-country study coordinated by WHO between 2011 and 2016.
While the findings of this study are encouraging, WHO continues to highlight the urgent need for new and improved tools in the global response to malaria. To prevent an erosion of the impact of core vector control tools, WHO also underscores the critical need for all countries with ongoing malaria transmission to develop and apply effective insecticide resistance management strategies.
Diagnosis and treatment
Early diagnosis and treatment of malaria reduces disease and prevents deaths. It also contributes to reducing malaria transmission. The best available treatment, particularly for P. falciparum malaria, is artemisinin-based combination therapy (ACT).
WHO recommends that all cases of suspected malaria be confirmed using parasite-based diagnostic testing (either microscopy or rapid diagnostic test) before administering treatment. Results of parasitological confirmation can be available in 30 minutes or less. Treatment, solely on the basis of symptoms should only be considered when a parasitological diagnosis is not possible. More detailed recommendations are available in the third edition of the “WHO Guidelines for the treatment of malaria”, published in April 2015.
Antimalarial drug resistance
Resistance to antimalarial medicines is a recurring problem. Resistance of P. falciparum malaria parasites to previous generations of medicines, such as chloroquine and sulfadoxine-pyrimethamine (SP), became widespread in the 1950s and 1960s, undermining malaria control efforts and reversing gains in child survival.
Protecting the efficacy of antimalarial medicines is critical to malaria control and elimination. Regular monitoring of drug efficacy is needed to inform treatment policies in malaria-endemic countries, and to ensure early detection of, and response to, drug resistance.
In 2013, WHO launched the Emergency response to artemisinin resistance (ERAR) in the Greater Mekong subregion (GMS), a high-level plan of attack to contain the spread of drug-resistant parasites and to provide life-saving tools for all populations at risk of malaria. But even as this work was under way, additional pockets of resistance emerged independently in new geographic areas of the subregion. In parallel, there were reports of increased resistance to ACT partner drugs in some settings. A new approach was needed to keep pace with the changing malaria landscape.
At the World Health Assembly in May 2015, WHO launched the Strategy for malaria elimination in the Greater Mekong subregion (2015–2030), which was endorsed by all the countries in the subregion. Urging immediate action, the strategy calls for the elimination of all species of human malaria across the region by 2030, with priority action targeted to areas where multidrug resistant malaria has taken root.
With technical guidance from WHO, all countries in the region have developed national malaria elimination plans. Together with partners, WHO is providing ongoing support for country elimination efforts through the Mekong Malaria Elimination programme, an initiative that evolved from the ERAR
Surveillance
Surveillance entails tracking of the disease and programmatic responses, and taking action based on the data received. Currently, many countries with a high burden of malaria have weak surveillance systems and are not in a position to assess disease distribution and trends, making it difficult to optimize responses and respond to outbreaks.
Effective surveillance is required at all points on the path to malaria elimination. Stronger malaria surveillance systems are urgently needed to enable a timely and effective malaria response in endemic regions, to prevent outbreaks and resurgences, to track progress, and to hold governments and the global malaria community accountable.
In March 2018, WHO released a reference manual on malaria surveillance, monitoring and evaluation, monitoring and evaluation. The manual provides information on global surveillance standards and guides countries in their efforts to strengthen surveillance systems.
Elimination
Malaria elimination is defined as the interruption of local transmission of a specified malaria parasite species in a defined geographical area as a result of deliberate activities. Continued measures are required to prevent re-establishment of transmission. Malaria eradication is defined as the permanent reduction to zero of the worldwide incidence of malaria infection caused by human malaria parasites as a result of deliberate activities. Interventions are no longer required once eradication has been achieved.
Globally, the elimination net is widening, with more countries moving towards the goal of zero malaria. In 2019, 27 countries reported fewer than 100 indigenous cases of the disease, up from 6 countries in 2000.
Countries that have achieved at least 3 consecutive years of 0 indigenous cases of malaria are eligible to apply for the WHO certification of malaria elimination. Over the last two decades, 11 countries have been certified by the WHO Director-General as malaria-free: United Arab Emirates (2007), Morocco (2010), Turkmenistan (2010), Armenia (2011), Sri Lanka (2016), Kyrgyzstan (2016), Paraguay (2018), Uzbekistan (2018), Algeria (2019), Argentina (2019) and El Salvador (2021). The WHO Framework for malaria elimination (2017) provides a detailed set of tools and strategies for achieving and maintaining elimination. In January 2021, WHO published a new manual, Preparing for certification of malaria elimination, with extended guidance for countries that are approaching elimination or preparing for elimination certification.
Vaccines against malaria
RTS,S/AS01 (RTS,S) is the first and, to date, the only vaccine to show that it can significantly reduce malaria, and life-threatening severe malaria, in young African children. It acts against P. falciparum, the most deadly malaria parasite globally and the most prevalent in Africa. Among children who received 4 doses in large-scale clinical trials, the vaccine prevented approximately 4 in 10 cases of malaria over a 4-year period.
In view of its public health potential, WHO’s top advisory bodies for malaria and immunization have jointly recommended phased introduction of the vaccine in selected areas of sub-Saharan Africa. Three countries – Ghana, Kenya and Malawi – began introducing the vaccine in selected areas of moderate and high malaria transmission in 2019. Vaccinations are being provided through each country’s routine immunization programme.
The pilot programme will address several outstanding questions related to the public health use of the vaccine. It will be critical for understanding how best to deliver the recommended 4 doses of RTS,S; the vaccine’s potential role in reducing childhood deaths; and its safety in the context of routine use.
This WHO-coordinated programme is a collaborative effort with Ministries of Health in Ghana, Kenya and Malawi and a range of in-country and international partners, including PATH, a non-profit organization, and GSK, the vaccine developer and manufacturer.
Financing for the vaccine programme has been mobilized through a collaboration between 3 major global health funding bodies: Gavi, the Vaccine Alliance, the Global Fund to Fight AIDS, Tuberculosis and Malaria, and Unitaid.
WHO response
WHO Global technical strategy for malaria 2016-2030
The WHO Global technical strategy for malaria 2016-2030 – adopted by the World Health Assembly in May 2015 – provides a technical framework for all malaria-endemic countries. It is intended to guide and support regional and country programmes as they work towards malaria control and elimination.
The Strategy sets ambitious but achievable global targets, including:
- reducing malaria case incidence by at least 90% by 2030;
- reducing malaria mortality rates by at least 90% by 2030;
- eliminating malaria in at least 35 countries by 2030;
- preventing a resurgence of malaria in all countries that are malaria-free.
This Strategy was the result of an extensive consultative process that spanned 2 years and involved the participation of more than 400 technical experts from 70 Member States.
The Global Malaria Programme
The WHO Global Malaria Programme coordinates WHO’s global efforts to control and eliminate malaria by:
- setting, communicating and promoting the adoption of evidence-based norms, standards, policies, technical strategies, and guidelines;
- keeping independent score of global progress;
- developing approaches for capacity building, systems strengthening, and surveillance; and
- identifying threats to malaria control and elimination as well as new areas for action.
The Programme is supported and advised by the Malaria Policy Advisory Committee (MPAC), a group of global malaria experts appointed following an open nomination process. The mandate of MPAC is to provide strategic advice and technical input, and extends to all aspects of malaria control and elimination, as part of a transparent, responsive and credible policy-setting process.
“High burden high impact approach”
At the World Health Assembly in May 2018, the WHO Director-General, Dr Tedros Adhanom Ghebreyesus, called for an aggressive new approach to jump-start progress against malaria. A new country-driven response – “ High burden to high impact” – was launched in Mozambique in November 2018.
The approach is currently being driven by the 11 countries that carry a high burden of the disease (Burkina Faso, Cameroon, Democratic Republic of the Congo, Ghana, India, Mali, Mozambique, Niger, Nigeria, Uganda and United Republic of Tanzania). Key elements include:
- political will to reduce the toll of malaria;
- strategic information to drive impact;
- better guidance, policies and strategies; and
- a coordinated national malaria response.
Catalysed by WHO and the RBM Partnership to End Malaria, “High burden to high impact” builds on the principle that no one should die from a disease that can be prevented and diagnosed, and that is entirely curable with available treatments.













 Posted by Ed Darrell
Posted by Ed Darrell